|
Precipitation reactions are usually represented solely by net ionic equations. Precipitate is what falls out of a super saturated solution or what remains after a super-saturated solution evaporates. A net ionic equation must be balanced on both sides not only in terms of atoms of elements, but also in terms of electric charge. The net ionic equation only shows the precipitation reaction. Since they go through the equation unchanged, they can be eliminated to show the net ionic equation: These are called spectator ions because they remain unchanged throughout the reaction. An example of a precipitation reaction is given below: CdSO 4(aq) K 2S(aq) CdS(s) K 2SO 4(aq) Both reactants are aqueous and one product is solid. In the equation above, A and D - ions are present on both sides of the equation. The resulting equation looks like that below:Ī ( aq ) B -( aq ) C ( aq ) D - ( aq ) → A ( aq ) D - ( aq ) C B( s ) Precipitates do not dissociate in water, so the solid should not be separated. The first step to writing a net ionic equation is to separate the soluble (aqueous) reactants and products into their respective cations and anions. Because this particular reaction is a precipitation reaction, states of matter can be assigned to each variable pair:Ī B( aq ) C D( aq ) → A D( aq ) C B( s ) To understand the definition of a net ionic equation, recall the equation for the double replacement reaction. Many reactions of this type involve the exchange of ions between ionic compounds in aqueous solution and are sometimes referred to as double displacement. In the example above Na (aq) and NO3- are present as both products and reactants. of 6. Net ionic equations are found by writing the full equation and then. How many grams of precipitate form and this reaction produces 18.7 g precipitate. If all the ions in a reaction are shown to be soluble, then no precipitation reaction occurs. Precipitates and Calculations A chemist combines 300 mL of a 0.3 M solution with 200 mL of 0.4 M solution. If an ion is insoluble based on the solubility rules, then it forms a solid with an ion from the other reactant. A double displacement reaction is also called a double replacement. The chemical bonds between the reactants may be either covalent or ionic. Usually, a double displacement reaction results in precipitate formation. If the rules state that an ion is soluble, then it remains in its aqueous ion form. A double displacement reaction is a type of chemical reaction in which the reactant ions exchange places to form new products. Sulfides formed with group 2 cations and hydroxides formed with calcium, strontium, and barium are exceptions. 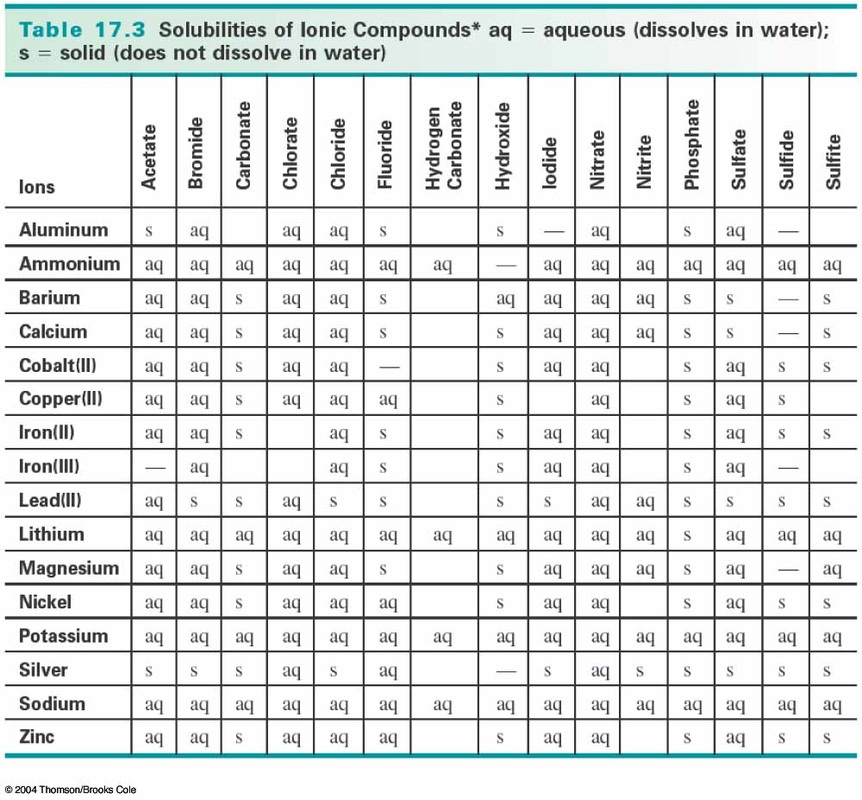
\)), sulfides, oxides, and hydroxides (\(OH^-\)) are insoluble.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
